Standard
S1
S2
S3
S4
S5
S6
Concentration (ng / ml)
0
0.09
0.78
3.12
25
200
STORAGE
1. Unopened test kits should be stored at 2-8? C upon receipt and the microtiter plate should be kept in a sealed bag to minimize exposure to damp air. The test kit may be used throughout the expiration date of the kit. Refer to the package label for the expiration date.
2. Opened test kits will remain stable until the expiring date shown, provided it is stored as prescribed above.
3. A microtiter plate reader with a ba-n-dwidth of 10 nm or less and an optical density range of 0-3 OD or greater at 450nm wavelength is acceptable for use in absorbance measurement.
REAGENT PREPARATION
Bring all reagents to room temperature before use.
1. Wash Buffer If crystals have formed in the concentrate, warm up to room temperature and mix gently until the crystals have completely dissolved. Dilute 20 ml of Wash Buffer Concentrate into deionized or distilled water to prepare 500 ml of Wash Buffer.
Precaution: The Stop Solution provided with this kit is an acid solution. Wear eye, ha-nd, face, and clothing protection when using this material.
OTHER SUPPLIES REQUIRED
1 Microplate reader capable of measuring absorbance at 450 nm, with the correction wavelength set at 540 nm or 570 nm.
2 Pipettes and pipette tips.
3 Deionized or distilled water.
4 Squirt bottle, manifold dispenser, or automated microplate washer.
5 Serum Use a serum separator tube (SST) and allow samples to clot for 30 minutes before centrifugation for 15 minutes at 1000 g. Remove serum and assay immediately or aliquot and store samples at -20 ° C. Avoid repeated freeze-thaw cycles.
6 Plasma Collect plasma using citrate, EDTA, or heparin as an anticoagulant. Centrifuge for 15 minutes at 1000 g within 30 minutes of collection. Assay immediately or aliquot and store samples at -20 ° C. Avoid repeated freeze-thaw cycles.
SAMPLE COLLECTION AND STORAGE
Note: Grossly hemolyzed samples are not suitable for use in this assay.
SAMPLE PREPARTION
Recommend to dilute the serum or plasma samples with Sample Diluent (1: 5000) before test. The suggested 5000-fold dilution
can be achieved by adding 2μl sample to 98μl of Sample Diluent, Complete the 5000-fold dilution by adding 2μl of this solution to 198μl of Sample Diluent. The recommended dilution factor is for reference only. The optimal dilution factor should be determined by users according to their particular experiments.
ASSAY PROCEDURE
Bring all reagents and samples to room temperature before use. It is recommended that all samples, sta-n-dards, and controls be assayed in duplicate.
1 Add 50μl of Sta-n-dard, Sample per well. Add 50ul HRP-conjugate to each well immediately. Mix well with the pipette or shake the plate gently for 60 seconds.
2 Then incubate for 30 minutes at 37 ° C.
3 Aspirate each well and wash, Wash by filling each well with Wash Buffer (200μl) using a squirt bottle, multi-channel pipette, manifold dispenser or autowasher. Repeating the process for a total of five time washes. Complete removal of liquid at each step is essential to good performance. After the last wash, remove any remaining Wash Buffer by aspirating or decanting. Invert the plate and blot it against clean paper towels.
4 Add 90μl of TMB Substrate to each well. Incubate for 20 minutes at 37 ° C. Keeping the plate away from drafts and other temperature fluctuations in the dark.
5 Add 50μl of Stop Solution to each well when the last four wells containing the lowest concentration of sta-n-dards develop obvious blue color. If color change does not appear uniform, gently tap the plate to ensure thorough mixing.
6 Determine the optical density of each well within 15 minutes, using a microplate reader set to 450 nm.
CALCULATION OF RESULTS
Average the duplicate readings for each sta-n-dard, control, and sample and divide the average zero sta-n-dard optical density. Create a sta-n-dard curve by reducing the data using computer software. As an alternative, construct a sta-n-dard curve by plotting the absorbance ratio for each sta-n-dard on the x-axis against the concentration on the y-axis and draw a best fit curve through the points on the graph. The data may be linearized by plotting the human IgG4 concentrations versus the ratio and the best fit line can be determined by regression analysis. This procedure will produce an adequate but less precise fit of the data. If samples have been diluted, the concentration read from the sta-n- dard curve must be multiplied by the dilution factor.
LIMITATIONS OF THE PROCEDURE
1 The kit should not be used beyond the expiration date on the kit label.
2 Do not mix or substitute reagents with those from other lots or sources.
3 It is important that the Calibrator Diluent selected for the sta-n-dard curve be consistent with the samples being assayed.
4 If samples generate values ​​higher than the highest sta-n-dard, dilute the samples with the appropriate Calibrator Diluent and repeat the assay.
5 Any variation in Sta-n-dard Diluent, operator, pipetting technique, washing technique, incubation time or temperature, and kit age can cause variation in binding.
6? This assay is designed to eliminate interference by soluble receptors, binding proteins, and other factors present in biological samples. Until all factors have been tested in the Immunoassay, the possibility of interference cannot be excluded. TECHNICAL HINTS
7 Centrifuge vials before opening to collect contents.
8 When mixing or reconstituting protein solutions, always avoid foaming.
9 To avoid cross-contamination, change pipette tips between additions of each sta-n-dard level, between sample additions, and between reagent additions. Also, use separate reservoirs for each reagent.
10 When using an automated plate washer, adding a 30 second soak period following the addition of wash buffer, and / or rotating the plate 180 degrees between wash steps may improve assay precision.
11 To ensure accurate results, proper adhesion of plate sealers during incubation steps is necessary.
12 Substrate Solution should remain colorless until added to the plate. Keep Substrate Solution protected from light. Substrate Solution should change from colorless to gradations of blue.
13 Stop Solution should be added to the plate in the same order as the Substrate Solution. The color developed in the wells will turn from blue to yellow upon addition of the Stop Solution. Wells that are green in color indicate that the Stop Solution has not mixed thoroughly with the Substrate Solution.
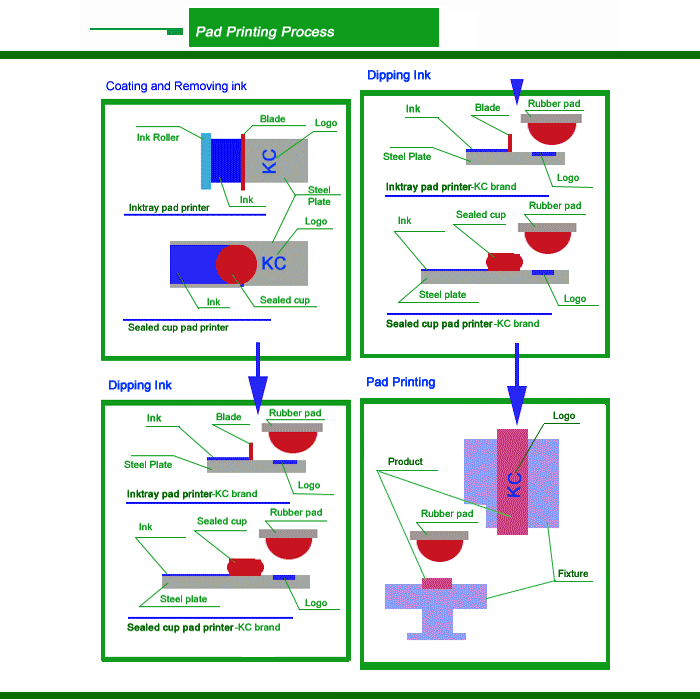 The oiled printing machine is the representative work of environmental protection tendency, which makes the VOC volatilization of the open ink transfer printing machine reduced by 95%. In Europe, the mainstream market is dominated by the printing machine, the open printing machine is only in eastern Europe, the cis countries have a certain distribution. In the long run, the oil-cup printing machine will spread to Asia, America, and Africa, and will be popularized in some large enterprises. However, the printing speed of the printing machine is challenging, it is difficult to manufacture, and the friction resistance of the oil cup and steel plate is a thorny problem. To explore from a perspective of printing pad printing machine, the cleaning device is an obvious progress, theory proves that the glue head can only transfer 67% of the ink, while 33% of ink will remain on the surface glue head into the next cycle, the viscous ink, some residual ink will re-enter the ink link, and part to remain in the rubber surface hinder the transfer of ink, in the first period of aging, the phenomenon is more serious, the cleaning device can will remain on the surface of the rubber head, 93% of ink wash, reduces the impact of a print link.
The oiled printing machine is the representative work of environmental protection tendency, which makes the VOC volatilization of the open ink transfer printing machine reduced by 95%. In Europe, the mainstream market is dominated by the printing machine, the open printing machine is only in eastern Europe, the cis countries have a certain distribution. In the long run, the oil-cup printing machine will spread to Asia, America, and Africa, and will be popularized in some large enterprises. However, the printing speed of the printing machine is challenging, it is difficult to manufacture, and the friction resistance of the oil cup and steel plate is a thorny problem. To explore from a perspective of printing pad printing machine, the cleaning device is an obvious progress, theory proves that the glue head can only transfer 67% of the ink, while 33% of ink will remain on the surface glue head into the next cycle, the viscous ink, some residual ink will re-enter the ink link, and part to remain in the rubber surface hinder the transfer of ink, in the first period of aging, the phenomenon is more serious, the cleaning device can will remain on the surface of the rubber head, 93% of ink wash, reduces the impact of a print link.